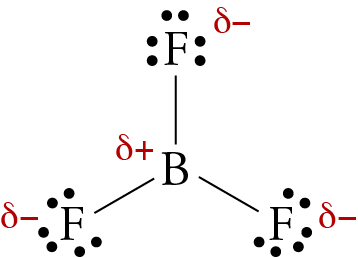
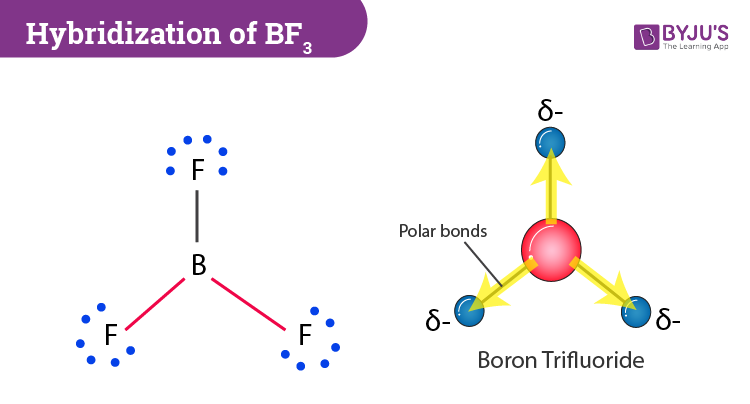
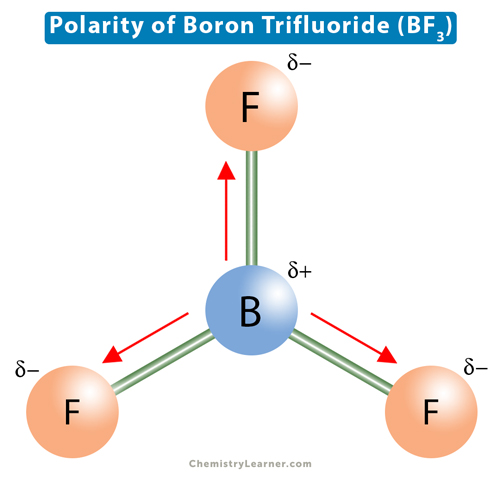
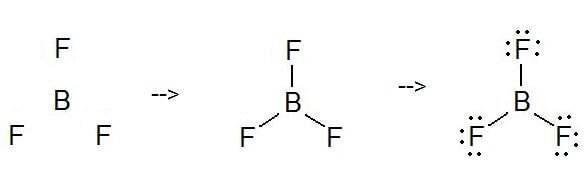
Ammonia (NH3) is a polar molecule while boron trifluoride (BF3), is a nonpolar molecule. What is the difference in the polarity of these compounds? - Quora

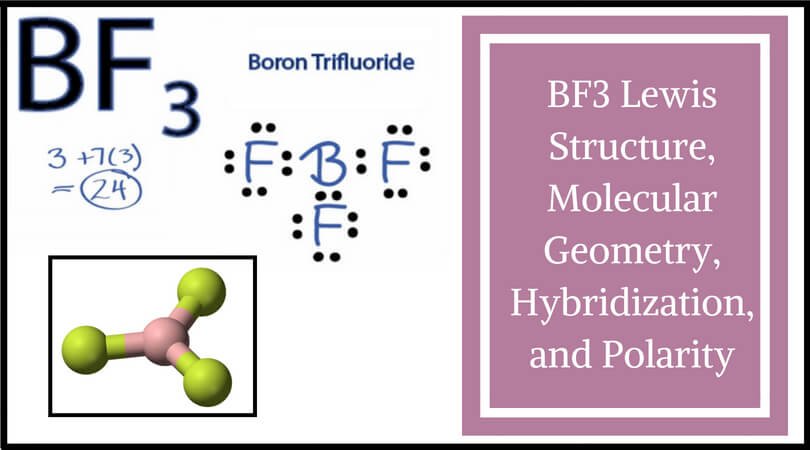
BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is non - polar. This is because :

Which of the following is a false statement about BF3? A. BF3 has trigonal planar molecular geometry. B. BF3 has trigonal pyramidal electronic geometry. C. All three bond angles in BF3 are

BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) | BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) Today in this video, we help you determine the molecular geometry of a
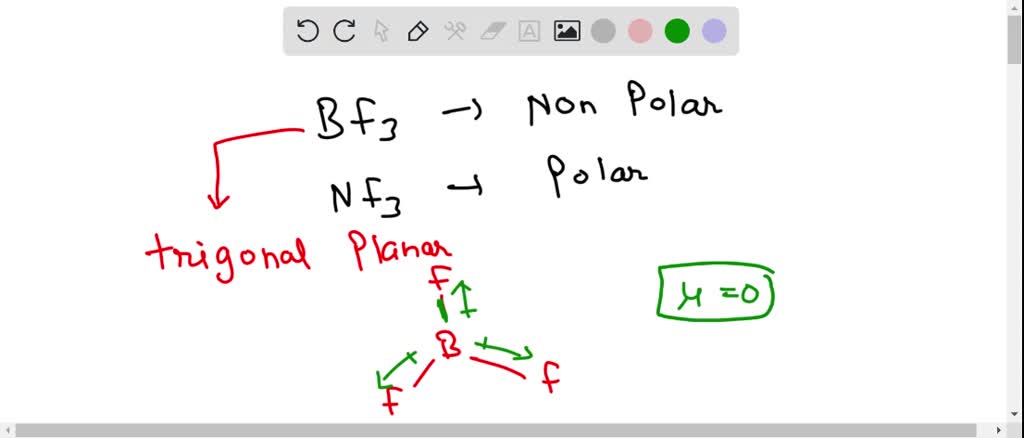
SOLVED: 41. The BF3 molecule is nonpolar; whereas the NF: molecule is polar: Which of the following statements accounts for the difference in polarity of the two molecules? In NFs, each F